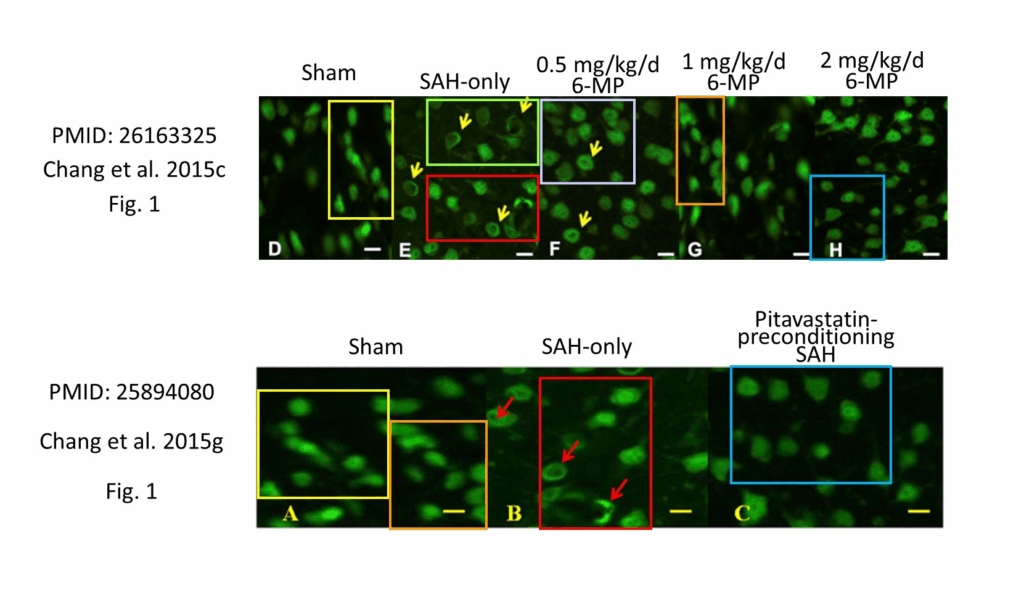
Annotated images: PubPeer
More than 200 papers on ways to prevent brain injury after a stroke contain problematic images, according to an analysis published today in PLOS Biology. Researchers found dozens of duplicated Western blots and reused images of tissues and cells purportedly showing different experimental conditions — both within a single paper and across separate publications.
As we reported last year, René Aquarius and Kim Wever, of the Radboud University Medical Center in the Netherlands, first noticed these patterns in 2023 when they started working on a systematic review of animal studies in the field. They had wanted to identify promising interventions for preventing early brain injury following hemorrhagic stroke. Instead, their efforts turned into an audit of suspicious papers in their field.
Of the 608 studies they analyzed, more than 240, or 40 percent, contained problematic images. So far, 19 of those articles have been retracted and 55 corrected, mostly from the researchers’ efforts to alert journals and publishers about the issues. Almost 90 percent of the problematic papers had a corresponding author based in China, and many appeared in major journals such as Stroke, Brain Research and Molecular Neurobiology.
Continue reading Widespread image reuse, manipulation uncovered in animal studies of brain injury


 Are there instances when similarities between papers should be fixed by a correction, rather than a retraction?
Are there instances when similarities between papers should be fixed by a correction, rather than a retraction?


