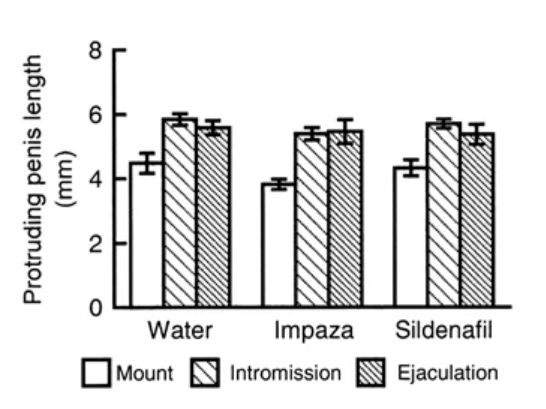
In January 2022, motivated by our experience with eClinicalMedicine, we wrote about mishandling of published errors by journal editors. We had noticed that the methods used for the analysis of a cluster randomized trial published in the journal were invalid. Using a valid approach, we reanalyzed the raw data, which were shared with us by the original authors. The trial’s results were overturned.
As Retraction Watch readers may recall, we subsequently submitted a manuscript describing why the original methods were invalid, what a valid analysis should be, and our results after conducting a valid analysis. After an initial desk rejection “in light of [the journal’s] pipeline” and further exchanges of correspondence, the journal shared our findings with the statistician involved in the original review and the original authors and sought their responses.
After receiving the responses, both of which we thought contained factually incorrect statements, the editorial team eventually suggested that we summarize our full manuscript as a 1000-word letter for submission to the journal. We did not agree that a letter would allow us to fully communicate our methods and reanalysis. Thus, to meet the journal’s word limit while fully laying out our arguments, we posted our additional points as a preprint and cited the preprint in a letter we submitted to the journal.
It was then that we met another roadblock to correcting the literature.
Continue reading When editors confuse direct criticism with being impolite, science loses


 For a host of reasons, a journal has retracted a paper co-authored by a researcher who reportedly once faced charges of practicing medicine without proper qualifications.
For a host of reasons, a journal has retracted a paper co-authored by a researcher who reportedly once faced charges of practicing medicine without proper qualifications. 
 When
When 