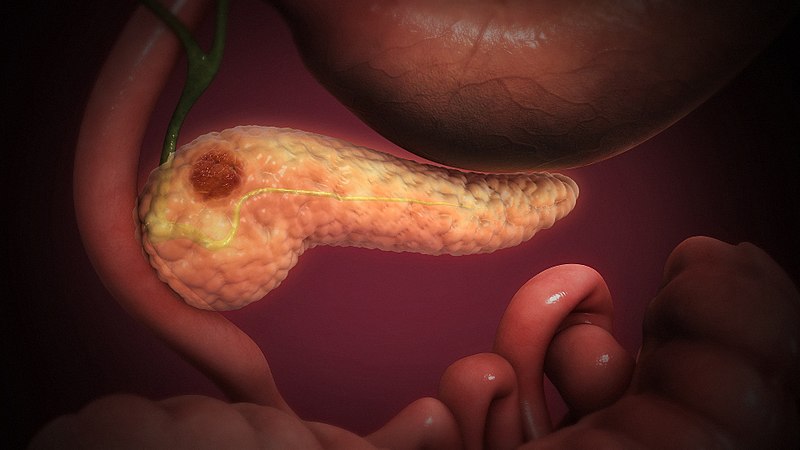
Researchers in China have lost a 2015 meta-analysis on pancreatic cancer, one of several retractions for members of the group stemming from a variety of abuses including bogus authorship and fake peer review.
The meta-analysis, “Correlation between serum levels of high mobility group box-1 protein and pancreatitis: a meta-analysis,” appeared in BioMed Research International, a Hindawi journal. The authors are affiliated with China Medical University in Shenyang and Shanghai Jiao Tong University.
Here’s the notice:
Continue reading Meta-meta-analysis meets with retraction for group that had faked peer review elsewhere


 For a host of reasons, a journal has retracted a paper co-authored by a researcher who reportedly once faced charges of practicing medicine without proper qualifications.
For a host of reasons, a journal has retracted a paper co-authored by a researcher who reportedly once faced charges of practicing medicine without proper qualifications.