 A paper in the Journal of Clinical Investigation has been retracted for manipulated images, including duplicated bands in a Western blot.
A paper in the Journal of Clinical Investigation has been retracted for manipulated images, including duplicated bands in a Western blot.
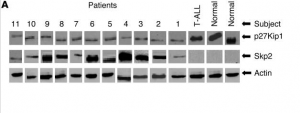
Take a look at figure one to the right and see if you can spot all the doubles.
Click here for a more zoomable image.
Here’s the notice:
The article “Loss of nuclear pro–IL-16 facilitates cell cycle progression in human cutaneous T cell lymphoma” is being retracted, with the agreement of the corresponding author. Several duplicated bands were recently identified in the immunoblot images in Figures 1 and 3, and the authors are unable to retrieve the original source files that were used to generate these data.
The paper has been cited nine times, according to Thomson Scientific’s Web of Knowledge.
Here’s Fig. 3, if you want to look there too (click here for better zoom):
We’ve asked the corresponding author and the editor for comment, and will update with anything we learn.
Hat tip: Steve McKinney
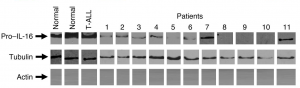
Could anyone point to the doubled lanes?
A couple more JCI papers with problematic data.
https://pubpeer.com/publications/A75065760963AF47B368F3AE3A2910
https://pubpeer.com/publications/B2BBD8918685C6E72AEB0344FDF329
Duplicated bands aside, how can a blot like this even be published in the first place? Resolution is terrible, and all bands are cut – how can you even compare two bands? Journals and editors should not accept this data, regardless of what reviewers might think of the paper.
“how can a blot like this even be published in the first place?” – what determines whether and where a paper is published often appears to be the names and affiliations of the authors, rather than the quality of the data.
Bingo.
Since it is still not obvious to me, which lane is duplicated??? Could anyone tell me the number of the lanes duplicated? – I sure agree that it is useless to show just cout out bands, but I thought any lane is duplicated.
In Fig. 1A, I’ve got p27Kip1 lane 1 and Skp2 lane 11 so far.
Actin for patients 5 and 11.
p27Kip1 for patient 6 looks to be a stretched version of p27Kip1 for patient 8, etc.
Thank you very much! I now found one questionable pair of bands as well: actin in patient 9 and T-ALL looks identical.
Just to add another couple, it appears to me that in Fig 1. “T-ALL p27Kip1” is just a slightly stretched resolution of “Skp2 Patient 6”, and in Fig 3. “Normal tubulin” is the 180 degree rotated version of “Patient 7 Pro-IL-16”.
Even if we ignore the band lane duplication, this figure is completely unacceptable as the resolution varies greatly across the panels, which alone indicates a great deal of sloppiness on the authors behalf. Its looks more like a JBC ’94 paper…
It’s more patchwork quilt than scientific figure.
I do fully concur with previous comments, how could this ever pass editors or even a peer review, never mind the manipulations? A mosaic of lanes, even the normalizer (actin)! What is the point of having this normalizer then? How can anyone seriously go on comparing anything here?
Who were the referees, just incompetent or simply friends of lead authors?
I suggest JCI apologizes for their embarrassing editorial procedure.
What’s scary is that “duplication” detected is truly lazy duplication (manipulation of a single stock image). What’s to stop someone from having an aliquot of /something/ and running it twice, let alone on two separate gels to create the illusion of difference?
Now I can’t sleep at night.
Exactly, the only lesson fraudsters take from this is to put more effort into faking. I actually believe in some labs more experimental effort is put into convincing faking than would go into honest science. But as long as the fake fits the sexy story, everyone will be happy.
And this PubPeer article too, very similar problems.
https://pubpeer.com/publications/4CD64087C332B7F037CE23D6AFBD99
I now agree that there are duplicated bands, and I agree that the resolution of the individual lanes/bands differs a lot – some of the bands are very pixeled.
But what does this tell about the journal (or science in general) – and especially the scientific value of all correct manuscript published? I fear that many manuscripts were not really checked, except if the last author is famous, or if a professors son is publishing as first author – then the peer-review system is just a confirmation that it would be OK to accept a manuscript for publication. But I also found published articles (in very different biophysics and neurobiology journals), which (in my view) lack any scientific training of the responsible author. I think it is hard for an editor to find a qualified reviewer – but usually it doesn’t matter since many customers of scientific journals are just the university libraries, which may not have real tools to check for the quality of science.
I do not know what this particularly egregious case tells us new about “science in general,” but with respect to this journal it seems fair to state that none of its articles should be believed. All articles in all journals should of course be evaluated with a critical eye, but such sloppy – and perhaps worse – performance by the editors, reviewers, and of course the authors indicates a journal-wide descent into irrelevance.
One “tool” that academics sometimes forget they possess is the ability to request that their institution’s library cancel subscriptions to suspect journals. In these days of budget cuts academic libraries are more than happy to trim costs by canceling journal subscriptions, and presented solely with this case a librarian wouldn’t exactly need to have their arm twisted off to give this journal the heave-ho.
In my experience, the university librarians are arm-twisted into subscribing to *bundles* of journals. In order to subscribe to some decent Elsevier journals. Our own library has to acquire the egregious ‘Medical Hypotheses’ as part of the package.
Then there are the variations in backgrounds — sometimes grainy, sometimes smooth. Some of the individual bands are truncated with a cleanly-sliced edge at the left or right or the bottom, with the truncated part replaced by a smooth grey spurious background.
It is as if the separate tessera of the mosaic came from different blots.
PPPR for the plant sciences has hit a serious stumbling block. The PPPR pool does not have sufficiently qualified individuals to detect the fine-scale details of band duplication and/or manipulation. One case was added to PubPeer, but without any supportive comments, or guidance, even though some concerns exist. If any RW commentators have any advice, since some of you have extremely astute observation skills, this would be welcome as PPPR moves forward with a deeper analysis of gels used in molecular analyses:
https://pubpeer.com/publications/DA95A526EEF49A72DC8D18CF202702#fb18051
Maybe we all should learn from this, that publishing scientific journals is a big market. Unfortunately, some of the decision makers easily judge popular research in the fancy journals like Science, Nature, Cell… as so much more important than unpublished findings or findings in not so mainstream, popular journals, that it is really bad for basic science to get even the attention of a decision maker.
Biochemistry. 2002 Dec 3;41(48):14306-12.
Prointerleukin-16 contains a functional CcN motif that regulates nuclear localization.
Wilson KC1, Cruikshank WW, Center DM, Zhang Y.
Author information
1The Pulmonary Center, Department of Medicine, Boston University School of Medicine, Boston, Massachusetts 02118, USA. [email protected]
https://pubpeer.com/publications/13C548B53D232E974DC22CB91FCE26#fb55058
Figures 5 and 6.
http://i.imgur.com/ZLlPjKm.jpg
Cell Immunol. 2005 Sep;237(1):17-27. Epub 2005 Nov 9.
Regulation of nuclear Prointerleukin-16 and p27(Kip1) in primary human T lymphocytes.
Wilson KC1, Cattel DJ, Wan Z, Rahangdale S, Ren F, Kornfeld H, Sullivan BA, Cruikshank WW, Center DM.
Author information
1The Pulmonary Center, Department of Medicine, Boston University School of Medicine, Boston, MA, USA.
https://pubpeer.com/publications/24114F49E602DA3DCE0BC391A23C0F#fb55057
Figure 2.
http://imgur.com/LUF6lX5
Figure 4.
http://imgur.com/h6AvQLj
Continuation Cell Immunol. 2005 Sep;237(1):17-27.
Figure 7.
http://imgur.com/LDYjMhO
Cell Immunol. 2005 Sep;237(1):17-27.
Figure 4 continued.
http://imgur.com/TRBdvqp
J Immunol. 2006 Feb 15;176(4):2337-45.
Chemokine receptor CXCR3 desensitization by IL-16/CD4 signaling is dependent on CCR5 and intact membrane cholesterol.
Rahangdale S1, Morgan R, Heijens C, Ryan TC, Yamasaki H, Bentley E, Sullivan E, Center DM, Cruikshank WW.
Author information
1Pulmonary Center, Boston University School of Medicine, Boston, MA 02118, USA.
Figure 8D
http://imgur.com/2Gso1wp
2019 ORI Case summary William Cruikshank.
https://ori.hhs.gov/content/case-summary-cruikshank-william-w